Presenting Kuopio Health members – FINBB
FINBB is building the world’s best biobank network
Finnish Biobanks – FINBB – aims to strengthen the competitiveness of Finnish health and biomedical research by making the sample and data resources of Finnish biobanks and their background organizations accessible to researchers. FINBB creates value for its members and owners by providing services both nationally and internationally.
FINBB is a cooperative owned by five wellbeing services counties, HUS, six universities, and Finnish Institute of Health and Welfare (THL). There are eight member biobanks maintained by its owners.
Since January 2020, FINBB has served as the national coordinator of the European biobank network BBMRI-ERIC. All ten Finnish biobanks participate in the network, including those outside FINBB’s ownership structure.
Towards a more unified biobank system
When FINBB was established, the goal was to increase coordination around biobank activities. Above all, there was a need to develop more harmonized operating models and facilitate researchers’ access to data. Finland wanted a clear single access route through which academic researchers as well as pharmaceutical and corporate actors could understand what data is available and how it can be utilized.
“The idea was not to create a new biobank, but to build an operator that brings together existing biobanks and their background organizations under one umbrella. Hospital biobanks focus on patient samples, while the biobanks of THL and the University of Oulu collect various population cohorts. This diverse entity offers researchers an exceptionally rich data foundation but requires a coordinating structure alongside it. FINBB was created to meet this need,” says FINBB’s CEO, Mervi Ahlroth, who has held the position since June 2025.

Mervi Ahlroth, FINBB’s CEO.
The cooperative model and the role of an intermediary
FINBB operates as a cooperative whose mission is to generate added value for its owners. The model works well in the context of biobank collaboration, as FINBB acts specifically as an intermediary: it does not sell samples or store data but connects research projects with biobanks and ensures that processes run smoothly.
“The same principle is reflected in how FINBB positions itself in legislative work. It is not a traditional advocacy organization, but it represents biobanks in various legislative and research law working groups and brings their perspectives into decision-making. This also relates to FINBB’s role in discussions around secondary use legislation and the European Health Data Space (EHDS), where the voice of biobanks needs to be clearly heard,” Ahlroth continues.
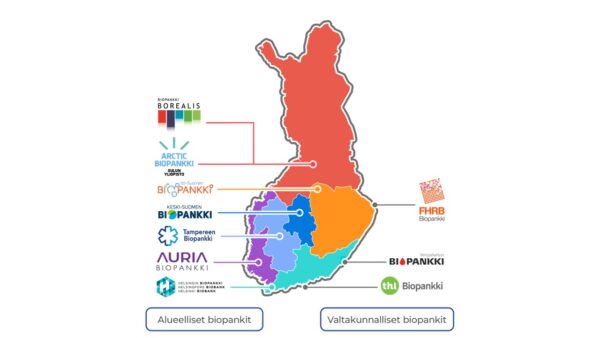
Finnish regional and national biobanks.
Fingenious® – A gateway to Finnish biobanks
FINBB’s digital service Fingenious® provides researchers with a single access point to all public biobanks in Finland. Through the service, both domestic and international researchers can search for existing cohorts, browse metadata, and conduct preliminary searches on what kind of biobank material is available in Finland.
Researchers can also submit feasibility and data access requests through Fingenious®, with FINBB’s experts supporting the process from start to finish.
“The service is not merely a self-service platform. Our experts support clients at every stage of the research process and assist, for example, in preparing research plans and permit applications. This comprehensive service is especially valued in international projects, where clients often return to Finland with new research topics,” Ahlroth explains.
Harmonized practices and accelerating research
One of FINBB’s key tasks is to harmonize biobank operating models as far as possible within different organizational systems. This work is being carried out, among others, in a development project funded by the Research Council of Finland (FIRI2024, National Research Infrastructure Roadmap 2025–2028), aiming to create shared models for process development and reduce overlapping work.
“FINBB also acts as a link to international research projects. Finland is one of the few countries with its own Biobank Act, which provides a competitive advantage. Biobanks can also invite sample donors to participate in new studies, which is not possible in all countries,” Ahlroth adds.
Biobanks as engines of knowledge and treatment development
Finnish biobank collections are significant on a global scale. The comprehensiveness and breadth of health data, the continuity of records, and the possibility of enriching data with registry information provide a considerable competitive edge internationally.
More than 14 million samples form an exceptional research resource, continuously generating new knowledge about disease mechanisms and enabling the development of personalized medicine. Biobanks can also collect new sample series according to researchers’ needs, further accelerating research.
Returning results derived from biobank samples to support patient care promotes early diagnosis and preventive treatment. For example, a pilot project launched in Helsinki and later expanded to other biobanks studied breast and ovarian cancer risk genes. Several participants were found to carry high-risk genetic variants they were previously unaware of. When information about elevated genetic risk was returned to participants through healthcare processes, they gained access to preventive care. Findings like these demonstrate the direct societal benefit of biobank activities.
“Finns remain highly research-positive, and social media-driven skepticism has not significantly affected consent rates. The challenge lies more in the resources of wellbeing services counties: more consents are obtained than samples can currently be collected and stored.”
The Fingenious® website also presents use case material demonstrating how biobank data has been utilized in research and what results have been achieved — from identifying genetic risk factors to advancing drug development, diagnostics, and healthcare effectiveness.
Data security and ethical responsibility
Data collected in biobanks is primarily processed and shared in pseudonymized form, meaning names and personal identity numbers are replaced with codes, and researchers accessing the data do not have access to identifying information. Identifiable data may only be shared in exceptional circumstances, for example when a researcher already holds prior research data based on participant consent and wishes to combine it with biobank data. Even then, the data is pseudonymized before analysis.
Pseudonymization protects participants’ privacy while preserving the ability to link new data and return research findings to participants if they so wish. Activities are supervised by Fimea, and legislation sets strict boundaries for handling samples and data. Finns’ strong trust in healthcare data protection is reflected in the high acceptance of biobank activities.
International networks and collaboration
FINBB plays an important role in the European BBMRI-ERIC (Biobanking and BioMolecular Resources Research Infrastructure — European Research Infrastructure Consortium Infrastructure) network as Finland’s national coordinator. Finnish experts participate in various working groups developing biobank quality, data, and operational practices. The network also provides access to EU project collaboration.
FINBB also participates in the EU-funded HealthHub Finland EDIH project, supporting small and medium-sized enterprises in developing health and wellbeing solutions. International pharmaceutical and health research companies form an important client group. They value the centralized Fingenious® service channel and the possibility of centralized contracting through FINBB, eliminating the need to negotiate separately with each biobank.
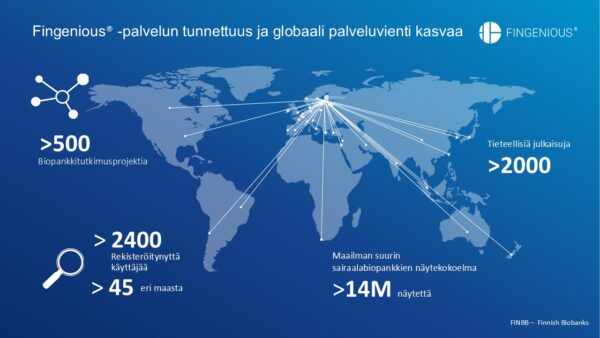
The reach of the Fingenious® service is extensive internationally.
Challenges and future opportunities
Biobanks are not statutory functions for wellbeing services counties, making them vulnerable to budget cuts. Securing stable core funding is therefore a key concern.
Emerging technologies such as omics, artificial intelligence, and data integration open new possibilities for facilitating clinical trial recruitment, generating deeper insights, and accelerating drug development. By strengthening coordination and communication across sectors, Finland could present itself internationally as a cohesive ecosystem offering high-quality samples, data analytics, and clinical research services through a coordinated national structure.
There is also growing discussion about reorganizing Finland’s health data infrastructure and defining FINBB’s future role within it. The aim is a more integrated system connecting biobanks and clinical research more seamlessly.
FINBB has already exported Finnish research services worth millions of euros, contributing to Finland’s national growth programme for health and wellbeing. The goal is to expand this activity further.
Kuopio Health membership
FINBB joined Kuopio Health in 2024 to become part of an active research and innovation environment.
“Kuopio has an inspiring and research-positive atmosphere. The city, region, university, and companies collaborate closely, creating fertile ground for partnerships and new initiatives. Through its expertise in biobank and research data, FINBB aims to bring significant added value to the ecosystem — strengthening Finland’s competitiveness and ensuring the country remains an international leader in the utilization of biobank samples and health data”, Ahloth ends.